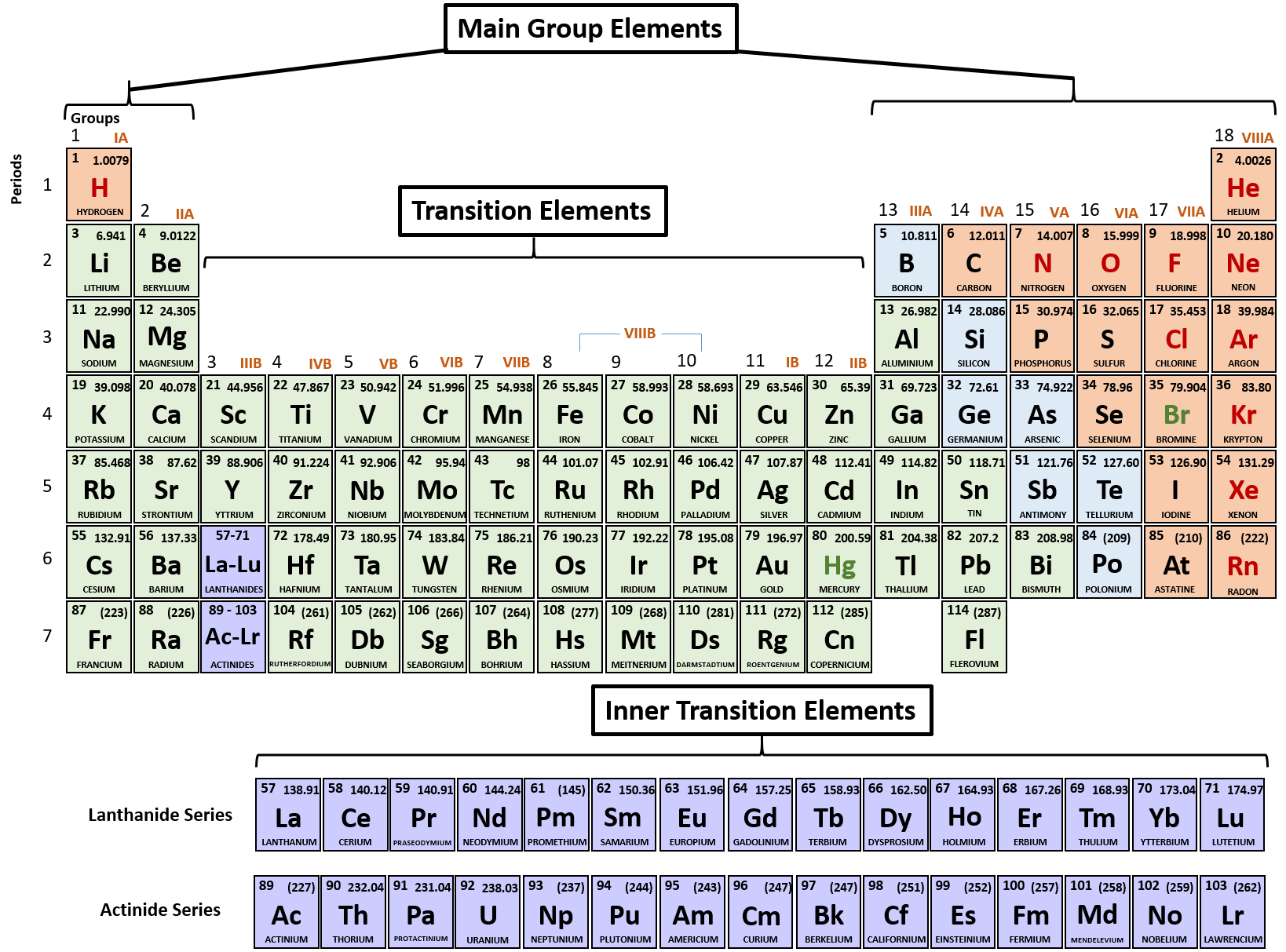
While your body can assemble many of the chemical compounds needed for life from their constituent elements, it cannot make elements. An element is a pure substance that is distinguished from all other matter by the fact that it cannot be created or broken down by ordinary chemical means. Elements and CompoundsĪll matter in the natural world is composed of one or more of the 92 fundamental substances called elements. A piece of cheese that weighs a pound on Earth weighs only a few ounces on the moon. In other words, weight is variable, and is influenced by gravity. An object of a certain mass weighs less on the moon, for example, than it does on Earth because the gravity of the moon is less than that of Earth. Where gravity strongly pulls on an object’s mass its weight is greater than it is where gravity is less strong. An object’s weight, on the other hand, is its mass as affected by the pull of gravity. An object’s mass is the amount of matter contained in the object, and the object’s mass is the same whether that object is on Earth or in the zero-gravity environment of outer space. An object’s mass and its weight are related concepts, but not quite the same. Scientists define matter as anything that occupies space and has mass. The substance of the universe-from a grain of sand to a star-is called matter. Explain how electrons occupy electron shells and their contribution to an atom’s relative stability.Identify the key distinction between isotopes of the same element.Distinguish between atomic number and mass number.Discuss the relationships between matter, mass, elements, compounds, atoms, and subatomic particles.Point to Remember: Mostly for transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core.By the end of this section, you will be able to: (See the above or below periodic table to see the valence electrons of inner transition metals). Inner transition elements can have valence electrons ranging from 3 to 16. In some inner transition metals, the electrons of incomplete d-orbitals are also considered as valence electrons. Hence, for inner transition elements, the electrons of both f-subshells as well as s-subshell behave like valence electrons. The inner transition elements have incomplete f- subshells and they are very close to the outer s-subshell. (See the above or below periodic table to see the valence electrons of transition metals).Īlso the two bottom rows at the bottom of the periodic table are the inner transition elements (or f-block elements) also have the similar case. Hence, the transition elements (i.e d-block elements from group 3 to 12) can have more valence electrons ranging from 3 to 12. So, the electrons of both d-subshell and s-subshell behave like valence electrons. It is more difficult to find the valence electrons of transition elements as they have incompletely filled d-subshell and this d-subshell is very close to the outer s-subshell. What about valence electrons of transition and inner transition elements?įor the transition elements and inner transition elements, the case is more complicated. 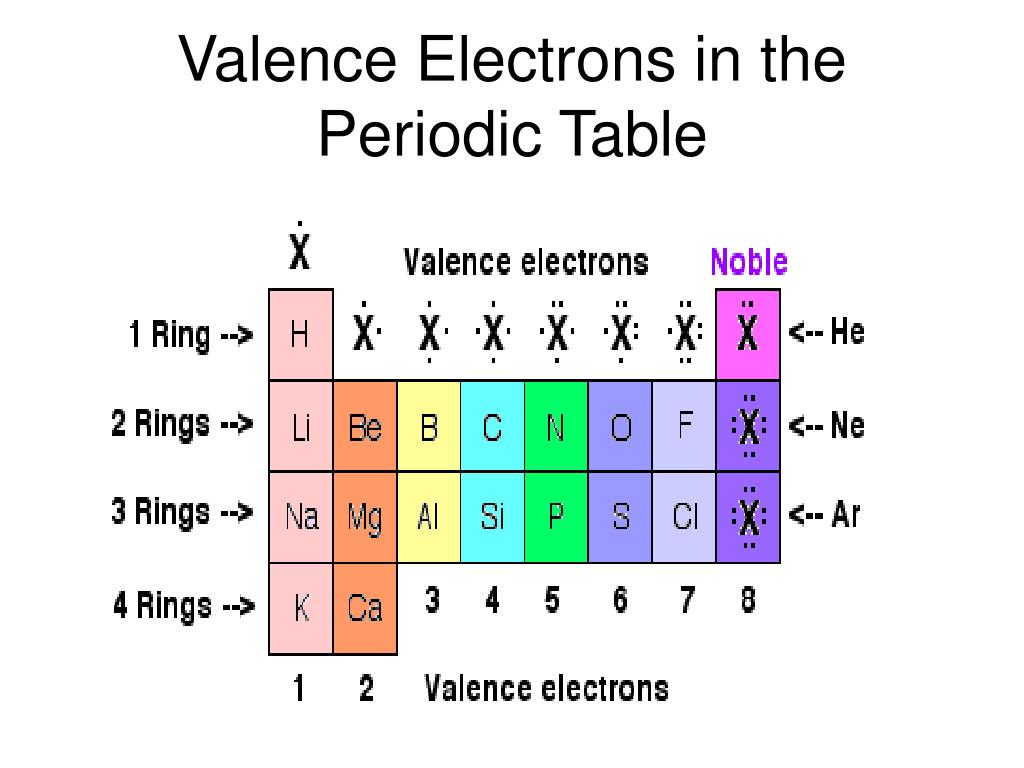
Well, this suits perfectly for the main group elements (i.e group 1, 2 and group 13 to 18), but what about the transition and inner transition elements? Hence, magnesium has 2 valence electrons. Here, you can see that the highest principal quantum number is 3, and the total electrons in this principal quantum number is 2. The electron configuration of magnesium is 1s 2 2s 2 2p 6 3s 2. Valence electrons can also be determined as the electrons present in the shell with highest principal quantum number (n). The magnesium element has 2 electrons in outermost orbit.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
